What is the difference between the hydrophilic heads and hydrophobic tails of the membrane?
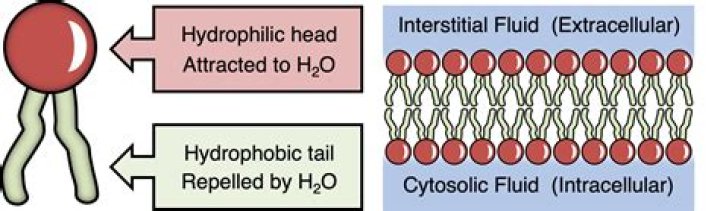
What is the difference between the hydrophilic heads and hydrophobic tails of the membrane?
The “head” of the molecule contains the phosphate group and is hydrophilic, meaning that it will dissolve in water. The “tail” of the molecule is made up of two fatty acids, which are hydrophobic and do not dissolve in water.
What is the head of a cell membrane?
What does hydrophilic head mean?
The hydrophilic heads attract water into the membrane and are then propelled away by the hydrophobic tails. Lastly the water is pulled across the membrane by the 2nd hydrophilic head. Hydrophilic means water loving and attracts water molecules while hydrophobic means water fearing and pushes water molecules away.
What is hydrophilic head?
The hydrophilic head is composed of a choline structure (blue) and a phosphate (orange). This is because they are two-faced molecules, with hydrophilic (water-loving) phosphate heads and hydrophobic (water-fearing) hydrocarbon tails of fatty acids.
How do hydrophilic and hydrophobic relate to the structure of a cell membrane?
When placed in water, hydrophobic molecules tend to form a ball or cluster. The hydrophilic regions of the phospholipids tend to form hydrogen bonds with water and other polar molecules on both the exterior and interior of the cell.
What is the hydrophobic head?
The phosphate group is the negatively-charged polar head, which is hydrophilic. The fatty acid chains are the uncharged, nonpolar tails, which are hydrophobic. Since the tails are hydrophobic, they face the inside, away from the water and meet in the inner region of the membrane.
What is the function of a hydrophobic?
Key terms
| Term | Meaning |
|---|---|
| Cell membrane | Specialized structure that surrounds the cell and its internal environment; controls movement of substances into/out of cell |
| Hydrophobic | Molecule that repels water (“water-fearing”) |
| Hydrophilic | Molecule that is attracted to water (“water-loving”) |
What does the hydrophilic part of the cell membrane do?
The hydrophilic heads of phospholipids in a membrane bilayer face outward, contacting the aqueous (watery) fluid both inside and outside the cell. Since water is a polar molecule, it readily forms electrostatic (charge-based) interactions with the phospholipid heads.
What is hydrophilic and hydrophobic?
Something defined as hydrophilic is actually attracted to water, while something that is hydrophobic resists water.
Is cell membrane hydrophobic or hydrophilic?
Thus, the membrane surfaces that face the interior and exterior of the cell are hydrophilic. In contrast, the interior of the cell membrane is hydrophobic and will not interact with water.
What does the hydrophobic tail do in the cell membrane?
Hydrophobic tails face inward and hydrophilic heads face outward. If you get these two ends mixed up, think of the root word “phobia” which means “fear.” Hydrophobic tails fear the water, so they will always try to be as far as possible from the water solutions in and out of the cell.
How does hydrophobic and hydrophilic relate to the cell membrane?
Plasma membranes range from 5–10 nm thick. Thus, both surfaces of the plasma membrane are hydrophilic (“water-loving”). In contrast, the interior of the membrane, between its two surfaces, is a hydrophobic (“water-hating”) or nonpolar region because of the fatty acid tails.



