What is the drug discovery process?
What is the drug discovery process?
The drug discovery process underpins the entire pharmaceutical industry, encompassing the early stages of research from target discovery and validation, right through to the identification of a drug candidate or lead compound. Initial identification of small therapeutic candidates comes about via a variety of streams.
What are the stages of preclinical trials?
Once a lead candidate is identified, a typical preclinical development program consists of six major efforts: manufacture of drug substance (DS)/active pharmaceutical ingredient (API); preformulation and formulation (dosage design); analytical and bioanalytical methods development and validation; metabolism and …
What are the three major phases of drug discovery?
Phases Stages
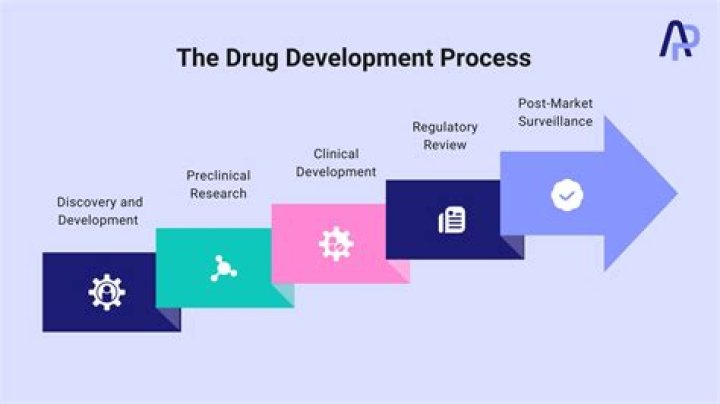
- Step 1: Discovery and Development.
- Step 2: Preclinical Research.
- Step 3: Clinical Development.
- Step 4: FDA Review.
- Step 5: FDA Post-market Safety Monitoring.
What happens in the preclinical stage of a drug trial?
In drug development, preclinical development, also termed preclinical studies or nonclinical studies, is a stage of research that begins before clinical trials (testing in humans) and during which important feasibility, iterative testing and drug safety data are collected, typically in laboratory animals.
What is drug discovery PDF?
Drug discovery is the process by which drugs are discovered and designed. It is a process which aims at identifying a compound therapeutically useful in curing & treating disease. Once a compound has shown its value in these tests, it will begin the process of drug development prior to clinical trials.
What are preclinical tests?
Preclinical testing is the link between drug discovery and availability to the patient. It takes at least 12–15 years for a drug to get from the lab to clinical use. The safety of a drug is established in toxicology studies, which are also used to establish biomarkers for monitoring.
What is the difference between non clinical and preclinical?
What do “preclinical” and “nonclinical” mean? “Pre” is Latin for “before” or “previous to,” while “non” is Latin for “negation” or “absence.” According to the Merriam-Webster dictionary, preclinical refers to studies occurring prior to clinical testing.
What is the significance of conducting preclinical stages tests in drug formulation and discovery?
The preclinical stage of drug development involves extensive testing in animal models to determine if the drug is safe for human trials and it if it performs as it should. Specifically, side effects of the drug need to be monitored and addressed.
What happens during preclinical drug trials?
Who is responsible for drug discovery?
Pharmaceutical scientists
Pharmaceutical scientists usually specialize in one aspect of the drug development process. They may: Design new drug therapies using natural or synthetic (man-made) ingredients. Uncover new ways to use existing drugs to treat different types of disease.



