What is the formation of carbocation?
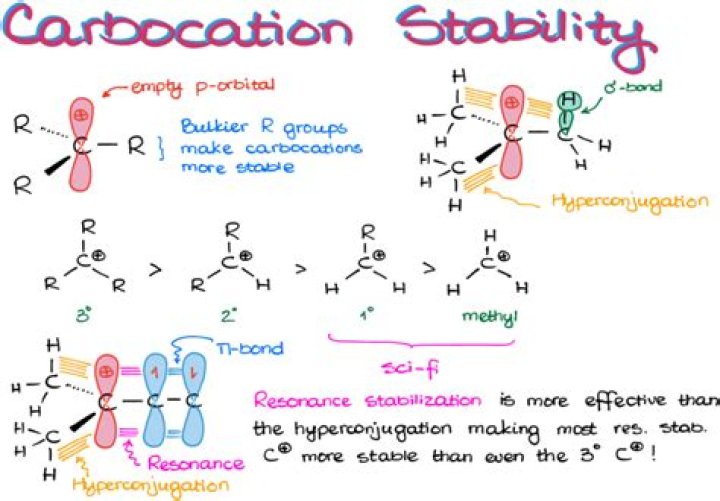
What is the formation of carbocation?
Cleavage of Bond of Carbon Whenever there is cleavage of the bond of carbon and atoms attached to it, the leaving group takes away the shared electrons. Thus leaving the carbon atom as electron deficient. As a result, a positive charge is developed forming a carbocation.
How do you rearrange a carbocation?
There are two types of carbocation rearrangements: a hydride shift and an alkyl shift. Once rearranged, the resultant carbocation will react further to form a final product which has a different alkyl skeleton than the starting material.
What reactions have Carbocations?
Examples of reactions involving carbocations: Eliminations via the E1 mechanism (Chapter 5) Electrophilic Additions to alkenes (Chapter 6) Electrophilic Additions to alkynes (Chapter 9)
How do you tell if a carbocation will rearrange?
When are rearrangements possible? If a secondary carbocation is vicinal to a tertiary carbon bearing a hydrogen, a 1,2- hydride shift should occur. If a secondary carbocation is vicinal to a quaternary carbon, a 1,2-alkyl shift should occur.
How is carbocation formed in sn1?
SN1 reaction mechanism follows a step-by-step process wherein first, the carbocation is formed from the removal of the leaving group. Then the carbocation is attacked by the nucleophile. Finally, the deprotonation of the protonated nucleophile takes place to give the required product.
What is carbocation give an example?
A carbocation is an ion with a positively-charged carbon atom. Among the simplest examples are methenium CH3+, methanium CH5+, and ethanium C2H7+. Some carbocations may have two or more positive charges, on the same carbon atom or on different atoms; such as the ethylene dication C2H42+.
How do you calculate carbocation?
The three factors that determine carbocation stability are adjacent (1) multiple bonds; (2) lone pairs; and (3) carbon atoms. An adjacent π bond allows the positive charge to be delocalized by resonance.
In which of the following rearranged 3 carbocation will not be formed?
In which of the following rearranged 3 degree carbocation will not be formed? The correct option is 1 as the cyclohexane ring is stable due to chair conformation. So it will not rearrange.
What is carbocation with example?
A carbocation (/ˌkɑːrboʊˈkætaɪən/) is an ion with a positively charged carbon atom. Among the simplest examples are the methenium CH + 3, methanium CH + 5 and vinyl C. 2H +
In which reaction mechanism carbocation is formed as an intermediate?
Both SN1 and E1 reactions feature carbocation intermediates. The first step in both reactions is the same: departure of the leaving group to form an intermediate carbocation. This is the rate determining step. It is a unimolecular reaction, and that accounts for the ” 1 ” in the names SN1 and E1 .
Do you know what a carbocation is?
How do you arrange carbocations in order of increasing stability?
Thus the observed order of stability for carbocations is as follows: tertiary > secondary > primary > methyl.