What is the freezing point depression constant of cyclohexane?
What is the freezing point depression constant of cyclohexane?
20.2 °C-kg per mol
The freezing point depression constant of cyclohexane is 20.2 °C-kg per mol of solute. The density and volume of cyclohexane are used to calculate the mass of the solvent.
What is the freezing point of cyclohexane and naphthalene solution?
13.87 The freezing point depression constants of the solvents cyclohexane and naphthalene are 20.1∘C/m and 6.94∘C/m, respectively. Which solvent would give a more accurate result if you are using freezing point depression to determine the molar mass of a substance that is soluble in either one?
What is the theoretical freezing point of cyclohexane?
6.50 °C.
Exercise: The freezing point of cyclohexane is 6.50 °C.
How do you calculate KF?
Divide the freezing point depression by the molal concentration so you have: Kf = delta Tf / cm. Insert the values for delta Tf and cm. For instance, if you have a solution with a molality of 0.455 which freezes at 3.17 degrees Celsius, then Kf would equal 3.17 divided by 0.455 or 6.96 degrees Celsius.
How do you determine freezing point?
Insert the thermometer in the slush, before the one you’re measuring turns completely liquid. Leave the thermometer in there until the point when it becomes all liquid. Write down the temperature when that happens. Make sure the thermometer you are using reads below 0 degree C.
How do you find the freezing point experiment?
In order to determine the freezing point of this pure solvent you must first heat it in a test tube to over 60 °C using a hot water bath, and then measure the temperature as a function of time as the liquid cools. At first the temperature will fall quite rapidly.
How do you find the freezing point of cyclohexane?
Exercise: The freezing point of cyclohexane is 6.50 °C. A solution is prepared by dissolving 0.5580g of an unknown solute in 33.50 g cyclohexane. The freezing point of the solution is 4.32 °C.
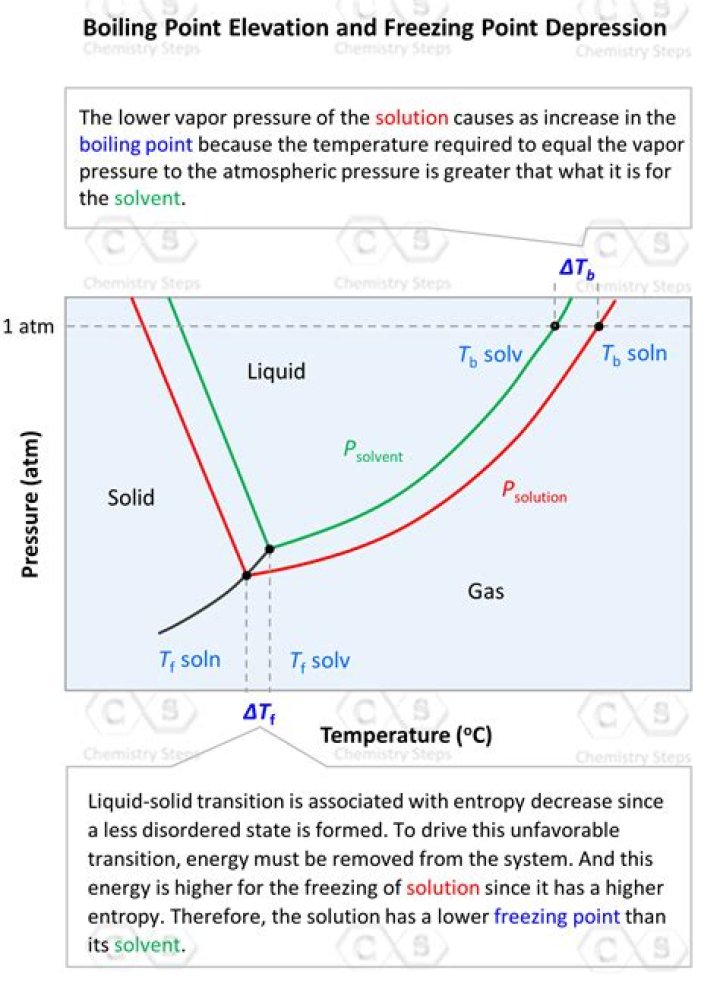
What is depression in freezing point How is molecular weight of a dissolved substance determined with this method?
Answer: Freezing point depression is a colligative property observed in solutions that results from the introduction of solute molecules to a solvent. The freezing points of solutions are all lower than that of the pure solvent and is directly proportional to the molality of the solute.
How do you calculate KF of water?
What is Beckmann method?
Beckmann thermometer is used to measure small temperature changes in the freezing point of pure solvent and solution. Beckmann thermometer is not used in determining the absolute value of freezing temperature of the solvent or that of the solution. It is therefore called a differential thermometer.