What is the latent heat of fusion in water?
What is the latent heat of fusion in water?
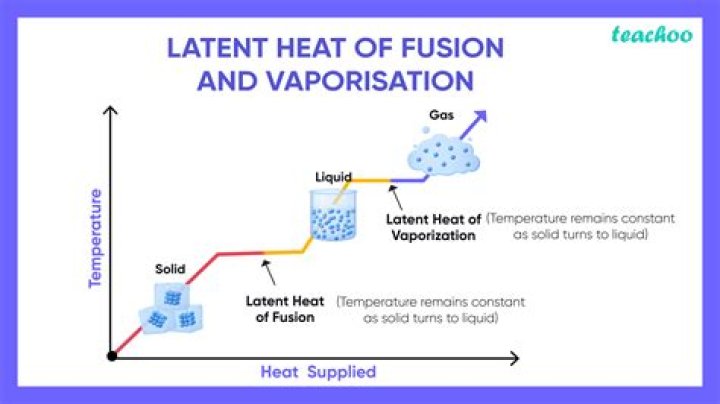
Similarly, while ice melts, it remains at 0 °C (32 °F), and the liquid water that is formed with the latent heat of fusion is also at 0 °C. The heat of fusion for water at 0 °C is approximately 334 joules (79.7 calories) per gram, and the heat of vaporization at 100 °C is about 2,230 joules (533 calories) per gram.
What is the latent heat of water in calories per gram?
Note: The latent heat of water at 0 degree Celsius for fusion is nearest to 334 joules per gram or 79.7 calories per gram. On the other hand, the latent heat of water at 100°C for vaporization is approximately 2230 joules per gram or 533 calories per gram.
How many J G is the heat of fusion of water?
Overview
| Substance | Heat of fusion | |
|---|---|---|
| (cal/g) | (J/g) | |
| water | 79.72 | 333.55 |
| methane | 13.96 | 58.99 |
| propane | 19.11 | 79.96 |
What is the latent heat of fusion in calories?
The latent heat of fusion for ice is 80 calories per gram (see calorie). This amount of heat is absorbed by each gram of ice in melting or is given up by each gram of water in freezing. The latent heat of vaporization of steam is 540 calories per gram, absorbed during vaporization or given up during condensation.
What is latent heat of fusion answer?
Answer: The latent Heat of Fusion is the change inenthalpy resulting from heating a given quantity of a substance to change its state from a solid to a liquid. The temperature at which this occurs is the melting point.
What is the formula for specific latent heat of fusion?
Specific Latent Heat of Fusion Formula The formula of specific latent heat of fusion is given by: Q = mL, which is the required specific latent heat of fusion equation.
What is specific latent heat of fusion?
The specific latent heat of fusion LF of a substance is the energy needed to change the state of 1kg of the substance from a solid to a liquid, at its melting point (without changing the temperature). 1 kg. 1 kg.
What is the value of latent heat of fusion?
For the material ice, it is defined as the amount of heat required to change one gram of ice to water at the same 00C. The value of latent heat of fusion of ice is 3.36×105JKg−1 in S.I. unit of system.
Why is it called latent heat of fusion?
Latent heat of fusion is the heat which is required to make 1kg of solid into liquid. This heat remains hidden until solid is completely transferred into liquid. It is called latent heat because latent means hidden.
What is meant by latent heat of fusion of ice is 80 cal GM?
When 1g o ice at a pressure of one atmosphere and at a temperature 0∘C is converted into 1 g of water, heat absorbed by the ice is 80 cal. …
What is the specific heat of water in calories?
4.184 joules
Because there are 4.184 joules in a calorie, the specific heat of water is 4.184 J/g-K. The ease with which a substance gains or loses heat can also be described in terms of its molar heat capacity, which is the heat required to raise the temperature of one mole of the substance by either 1oC or 1 K.
What is latent heat of fusion unit?
When a solid is converted into liquid at constant temperature (melting point of the substance) the amount of heat absorbed by it is called the latent heat of fusion. Heat is a form of energy . Hence , latent heat is expressed in units joule, erg, calorie or kilocalorie.