What is the mechanism of action of methyl salicylate formation?
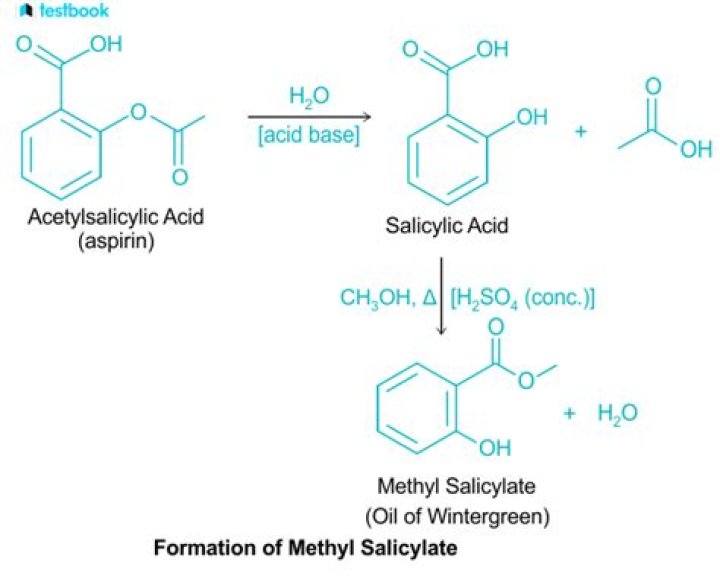
What is the mechanism of action of methyl salicylate formation?
Menthol also acts as a penetration enhancer, increasing the penetration of drugs when applied on the skin, to give a faster onset of action 6. Methyl salicylate is a salicylic acid derivative, and when combined with menthol, produces menthyl salicylate 6.
What are the roles of methyl salicylate in the synthesis of salicylic acid?
Methyl salicylate (an ester) can be hydrolyzed to produce salicylic acid. Acetic anhydride will be used in this experiment so that when ester is formed, water is not produced. As a result, the thermodynamic equilibrium will now be for the formation of an ester instead of its hydrolysis.
What is mechanism of action of aspirin?
Aspirin is non-selective and irreversibly inhibits both forms (but is weakly more selective for COX-1). It does so by acetylating the hydroxyl of a serine residue. Normally COX produces prostaglandins, most of which are pro-inflammatory, and thromboxanes, which promote clotting.
What is the difference between methyl salicylate and salicylic acid?
In methyl salicylate (oil of wintergreen), the COOH group of salicylic acid is esterified with methanol (CH3OH), whereas in acetylsalicylic acid (aspirin) the acid component of the ester is acetic acid, and salicylic acid contributes the phenolic ―OH group.
What is the role of methyl salicylate?
This product is used to treat minor aches and pains of the muscles/joints (such as arthritis, backache, sprains). Menthol and methyl salicylate work by causing the skin to feel cool and then warm. These feelings on the skin distract you from feeling the aches/pains deeper in your muscles and joints.
What functional group is methyl salicylate?
ester
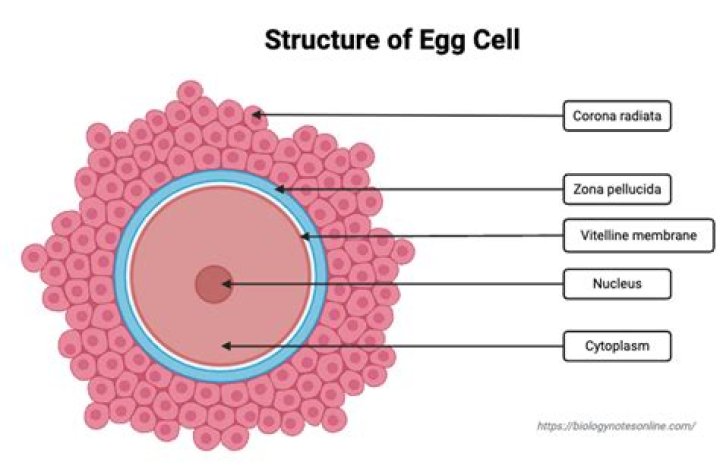
The difference in the two structures is a single functional group. In methyl salicylate (on the left) the benzene ring is substituted by two functional groups, a hydroxyl group (-OH) and an ester group (-COOCH3). In salicylic acid (on the right), the ester group has been replaced by a carboxylic acid group (-COOH).
What is the relationship between salicylic acid and aspirin?
This unique drug belongs to a family of compounds called the salicylates, the simplest of which is salicylic acid, the principal metabolite of aspirin. Salicylic acid is responsible for the anti-inflammatory action of aspirin, and may cause the reduced risk of colorectal cancer observed in those who take aspirin.
What drugs inhibit COX 1 and COX-2 enzymes?
6 Traditional NSAIDs, like Motrin (ibuprofen), aspirin, and Aleve (naproxen), while effective, can cause gastrointestinal problems including ulcers because they’re non-selective, meaning they inhibit both COX-1 and COX-2.
Is methyl salicylate the same as methanol?
Methyl salicylate (oil of wintergreen or wintergreen oil) is an organic compound with the formula C8H8O3. It is the methyl ester of salicylic acid. It is produced by many species of plants, particularly wintergreens. It is also produced synthetically, used as a fragrance and as a flavoring agent.
What is the product of methanol and salicylic acid?
When salicylic acid combines with methanol it becomes the ester known as methyl salicylate or oil of wintergreen.