What is the molecular geometry of AB3E2?
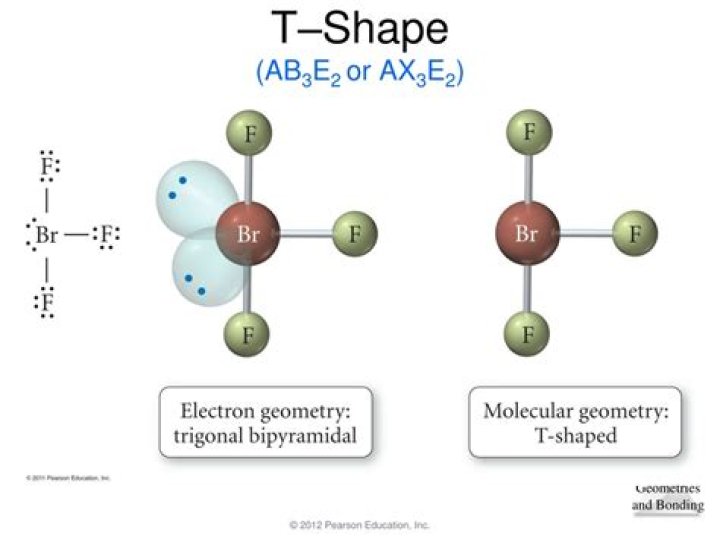
What is the molecular geometry of AB3E2?
For a molecule with a form of AB3E2 A B 3 E 2 , the A represents the central atom, the B represents the bonding pairs, and E represents the lone pairs. Molecules with this form have T-shaped molecular geometry whose parent molecular geometry is trigonal bipyramidal.
What is the shape of AB2E3 molecule?
trigonal bipyramidal shape
The AB2E3 A B 2 E 3 is trigonal bipyramidal shape.
What is the shape of AB2E2 type molecule?
The shape of AB2E2 is tetraheadle .
Why is ax3e2 T-shaped not trigonal planar?
This is an example of a t-shaped molecule. These molecules consist of three atoms surrounding a central atom. The lone pairs on the central atom repel the adjacent atoms, preventing the molecule from taking the shape of a trigonal planar molecule. …
Why are molecules of the form ab3e2 T-shaped and not trigonal planar?
Not only for E-E repulsion, you have to account for the repulsion between the lone pairs and the bonding pairs (aka E and B). The T-shaped would lead to a 120 degree between the two lone pairs and the B in the plane, and 90 degree between the plane (consists of two lone pairs and B) and two other Bs.
What is the shape of the ibr4 − ion?
octahedral
So, the electron geometry is octahedral.
What is the bond angle of AX3E2?
120o.
AX3E2. NOTES: This molecule is made up of 5 sp3d hybrid orbitals. Three orbitals are arranged around the equator of the molecule with bond angles of 120o.
Is ONCL trigonal planar?
Explanation: Now the electron pairs in the plane adopt a geometry that minimizes electronic interaction. And reasonably the electron pairs lie in a trigonal plane with respect to the other electron pairs. And so the electron pairs adopt a trigonal planar geometry.
What is the molecular shape of ONCL?
The molecular geometry of NOCl is bent (or angular) with an asymmetric charge distribution on the central atom.
What is the shape of ab3e type molecule?
AB3E: Ammonia, NH The domain geometry for a molecule with four electron pairs is tetrahedral, as was seen with CH 4 . In the ammonia molecule, one of the electron pairs is a lone pair rather than a bonding pair.