What is the molecular shape of H2CO3?
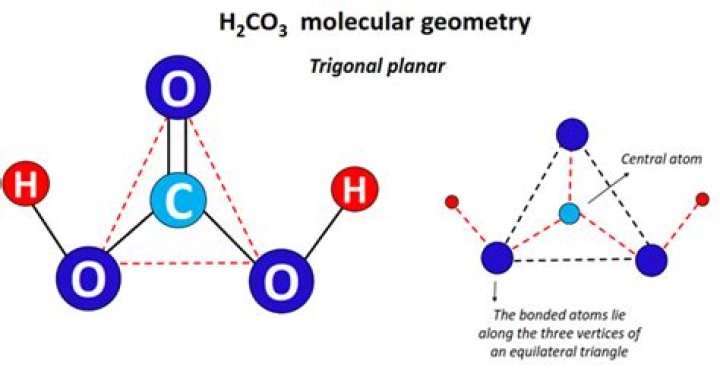
What is the molecular shape of H2CO3?
The geometry about the C atom is trigonal planar, but the C=O. dipole is not balanced by the C-O dipoles.
How do you write a Lewis structure for a molecule?
How to Draw a Lewis Structure
- Step 1: Find the Total Number of Valence Electrons.
- Step 2: Find the Number of Electrons Needed to Make the Atoms “Happy”
- Step 3: Determine the Number of Bonds in the Molecule.
- Step 4: Choose a Central Atom.
- Step 5: Draw a Skeletal Structure.
- Step 6: Place Electrons Around Outside Atoms.
Is Lewis structure a molecular formula?
Introduction: By counting valence electrons, we can predict the stable molecules that could form for a particular molecular formula. Molecular structures that are drawn using this formalism (of counting valence electrons) are called Lewis Structures.
What type of bond is H2CO3?
From the illustration provided above, it can be understood that the structure of carbonic acid consists of one carbon-oxygen double bond and two carbon-oxygen single bonds. The oxygen atoms participating in a single bond with the carbon each have one hydrogen atom attached to them.
Is H2CO3 a molecule?
carbonic acid, (H2CO3), a compound of the elements hydrogen, carbon, and oxygen. It is formed in small amounts when its anhydride, carbon dioxide (CO2), dissolves in water. Carbonic acid (H2CO3) is formed in small amounts when its anhydride, carbon dioxide… …
How many total atoms are in H2CO3?
The problem is, one molecule of H2CO3 contains six atoms, so is the question: “How many molecules?” or “How many atoms?” You need to clarify that, then you can answer the question.
Is structural formula the same as Lewis structure?
(3c) ·Lewis dot diagrams are used to represent valence electrons in an element. Structural formulas show the arrangements of atoms and bonds in a molecule and are represented by Lewis dot structures.
What is the molecular mass of H2CO3 with solution?
So the molecular mass of h2co3= 2+14+48 = 64 unit.. 2. Atomic mass of carbon is 14 hydrogen is 1 and oxygen is 16. So the molecular mass of c2h5oh = 28 + 5 + 16 + 1 = 50 unit..
Is H2CO3 an atom element or molecule?
Carbonic acid is a molecule which contains one carbon atom, three oxygen atom and two hydrogen atom. In the lewis structure of carbonic acid (H2CO3), carbon atom is the center atom and there are two -OH groups. Also, there is one double bond between carbon and oxygen atoms.