What is the NMR spectrum of ethanol?
What is the NMR spectrum of ethanol?
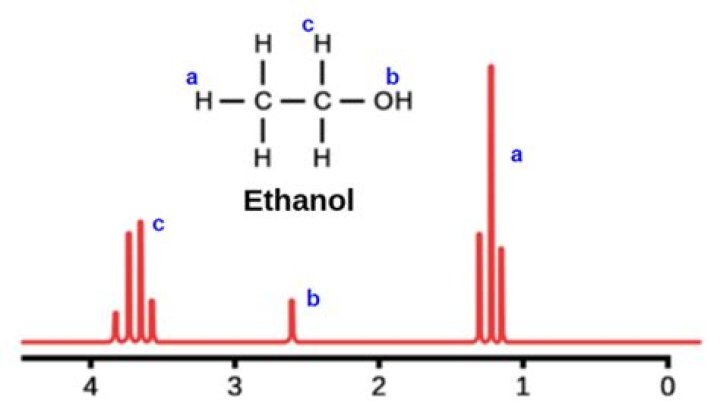
Ethanol 1 H NMR spectrum (high res.) If the spectrum of ethanol is recorded as a high-resolution spectrum, more detail is apparent and the peaks appear as singlets, doublets, triplets, quartets etc. The sets of peaks are due to interaction of protons from neighbouring groups.
How many NMR signals does ethanol have?
three NMR signals
Ethanol (CH3CH2OH), for example, gives three NMR signals, one of which is due to its OH proton.
Where does ethanol show up on NMR?
A reliable degree level organic chemistry text book quotes1. 0 – 5.0, but then shows an NMR spectrum for ethanol with a peak at about 6.1. The SDBS database (used throughout this site) gives the -OH peak in ethanol at about 2.6.
How NMR spectra are produced?
The sample is placed in a magnetic field and the NMR signal is produced by excitation of the nuclei sample with radio waves into nuclear magnetic resonance, which is detected with sensitive radio receivers. The alignment (polarization) of the magnetic nuclear spins in an applied, constant magnetic field B0.
What does NMR spectroscopy tell you?
Nuclear Magnetic Resonance (NMR) spectroscopy is an analytical chemistry technique used in quality control and research for determining the content and purity of a sample as well as its molecular structure. For example, NMR can quantitatively analyze mixtures containing known compounds.
Nuclear Magnetic Resonance Spectrum of Ethanol. The resonances are sensitive to the chemical environment of nonequivalent protons, an effect known as the chemical shift. A classic example is the ethanol molecule C C OH, which shows three chemically-distinct hydrogen atom sites, thus three NMR peaks with intensity ratios 3:2:1.
What does NMR stand for?
Nuclear Magnetic Resonance Spectrum of Ethanol. Nuclear magnetic resonance (NMR) spectroscopy can measure radio-frequency Zeeman transitions of proton spins in a magnetic field. It is more convenient to sweep the magnetic field through the resonances at a fixed frequency, typically 60 MHz. The resonances are sensitive to the chemical environment…
What is the history of NMR spectroscopy?
The first NMR spectrum of a fluid sample was taken of ethanol at Stanford University in 1951. Even from this early spectrum, the three hydrogen peaks can be resolved.
What is a singlet peak in NMR?
In NMR nomenclature, splitting of a peak into one is a singlet, two is a doublet, three is a triplet, four is a quartet, and so on. The first NMR spectrum of a fluid sample was taken of ethanol at Stanford University in 1951.