What is the product of the Friedel-Crafts reaction of benzene with 1 Chloropropane?
What is the product of the Friedel-Crafts reaction of benzene with 1 Chloropropane?
isopropyl benzene
When benzene is treated with $1 – $ chloropropane in presence of a lewis acid like aluminium trichloride, benzene undergoes friedel craft alkylation to give isopropyl benzene (also known as cummene) as a major product, and $n – $ propyl benzene as minor product.
What product is obtained from the reaction of 1 Chloropropane and benzene in the presence of Lewis acid?
For example, reaction of excess benzene with 1-chloropropane and aluminum chloride gives a good yield of isopropylbenzene (cumene).
Why do we get isopropyl benzene with 1 Chloropropane instead of N propylbenzene?
Therefore, isopropyl carbocation being more stable than n-propyl action, the former has a greater population than the latter. So, benzene later react more with isopropyl cation than n-propyl cation to produce more isopropyl benzene than n-propyl benzene.
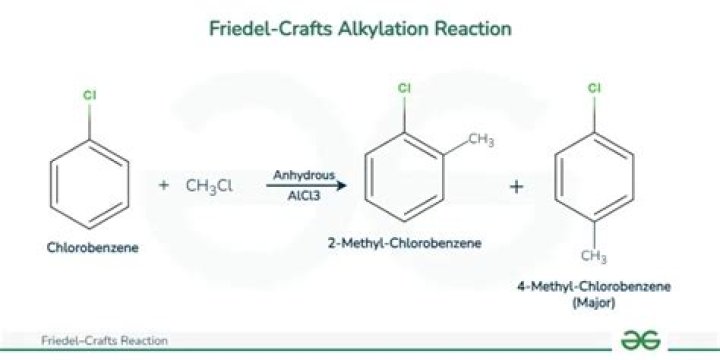
What is Friedel Craft alkylation reaction of benzene?
An alkyl group can be added to a benzene molecule by an electrophile aromatic substitution reaction called the Friedel‐Crafts alkylation reaction. The carbocation then reacts with the π electron system of the benzene to form a nonaromatic carbocation that loses a proton to reestablish the aromaticity of the system.
Which statement about the reaction of benzene with 1-chloropropane in presence of AlCl3 is incorrect?
Which statement about the reaction of benzene with 1-chloropropane in the presence of AlCl3 is incorrect? AlCl3 acts as a catalyst. Benzene undergoes electrophilic substitution. In the presence of AlCl3, 1-chloropropane reacts to give a carbenium ion.
What is the major product formed during the reaction of benzene with isopropyl alcohol in the presence of conc h2so4?
When benzene reacts with isopropyl alcohol, we get cumene as the major product.
Which statement about the reaction of benzene with 1 chloropropane in the presence of alcl3 is incorrect?
What are the possible products of the reaction between benzene and 2 Br₂ in the presence of an iron catalyst?
The reaction with bromine The reaction between benzene and bromine in the presence of either aluminum bromide or iron gives bromobenzene. Iron is usually used because it is cheaper and more readily available.
Which is Friedel Crafts reaction?
The Friedel–Crafts reactions are a set of reactions developed by Charles Friedel and James Crafts in 1877 to attach substituents to an aromatic ring. Friedel–Crafts reactions are of two main types: alkylation reactions and acylation reactions. Both proceed by electrophilic aromatic substitution.
Which is used as Friedel Crafts catalyst?
Anhydrous aluminium Chloride is used as catalyst in Friedel-crafts reaction.
Which statement about the reaction of benzene with cl2 in the presence of AlCl3 is in correct?
Correct Option: B The reaction between benzene and chlorine in the presence of a catalyst such as aluminium chloride gives Chlorobenzene. This reaction is an example of electrophilic substitution reaction since one of the hydrogen atoms on the ring is replaced by a chlorine atom.
What type of reaction are shown by benzene?
Substitution Reactions of Benzene and Other Aromatic Compounds
| Reaction Type | Typical Equation | |
|---|---|---|
| Halogenation: | C6H6 | + Cl2 & heat FeCl3 catalyst |
| Nitration: | C6H6 | + HNO3 & heat H2SO4 catalyst |
| Sulfonation: | C6H6 | + H2SO4 + SO3 & heat |
| Alkylation: Friedel-Crafts | C6H6 | + R-Cl & heat AlCl3 catalyst |