What is the purpose of saponification test?
What is the purpose of saponification test?
The saponification number depends on the molecular weight and the percentage concentration of fatty acid components present in FAMEs of oil. The SV is effectively used to determine the average relative molecular mass of oils and fats.
How do you explain saponification?
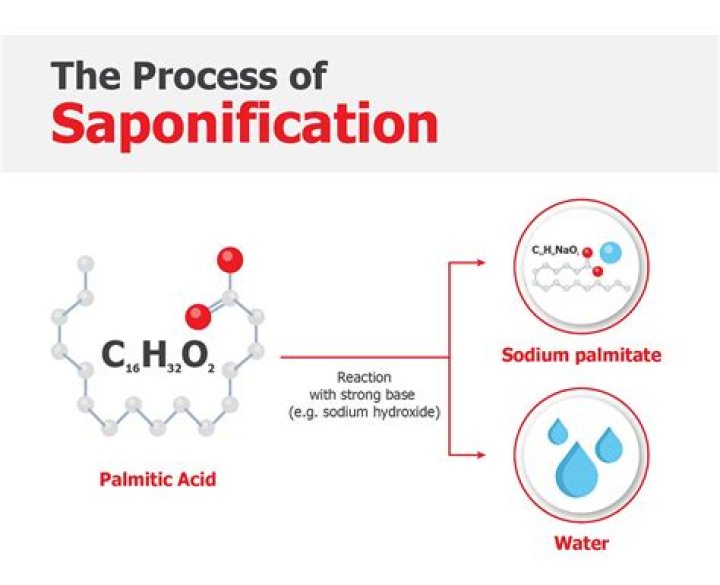
Saponification is a process by which triglycerides are reacted with sodium or potassium hydroxide (lye) to produce glycerol and a fatty acid salt called “soap.” The triglycerides are most often animal fats or vegetable oils. Using potassium hydroxide results in a soft soap.
What is saponification how it is prepared in laboratory explain?
Soaps. are sodium or potassium salts of long chain fatty acids. When triglycerides in fat/oil react with aqueous NaOH or KOH, they are converted into soap and glycerol. This is called alkaline hydrolysis of esters. Since this reaction leads to the formation of soap, it is called the Saponification .
What is the result of saponification?
As a result of the Saponification process, the fatty acids are hydrolyzed in presence of an alkali so as to form salts of alkali and alcohol. Upon cooling of the dissolved mixture, solid soap was observed the end of the process. A clear, pale pink solution indicates good results.
What is saponification value and its significance?
In addition, we also determine these for the verification of our raw materials and as apprenticeship tasks during training. Definition / Explanation. The saponification number is one of several characteristic values of fat. The saponification number indicates how much potassium hydroxide is needed to saponify 1g fat.
How is saponification value tested?
Saponification Value = (A – B) x N x 56.1 W This method is used to determine the total acid content, both free and combined, of tall oil. (Acid number only measures the free acid). The combined acids are primarily esters formed by reaction with the neutral components present in the original tall oil.
How do you know if saponification is complete?
The zap test is when you stick a bar of soap to your tongue. If it zaps you like a 9-volt battery, your soap is still not saponfied. If it doesn’t, it is probably done with the process. Again, saponification takes about 24-48 hours.
What affects saponification?
This study is executed for the factors that affect the process for saponification of J. curcas seed oil. D-optimal design was used to evaluate the effect of three factors, such as ethanolic KOH concentration, reaction temperature, and reaction time which were studied for the optimum saponification.
What does a low saponification value mean?
A high saponification value indicates that the sample has a shorter fatty acid chain and a lower molecular weight. A low saponification value indicates that the sample has a longer fatty acid chain and a higher molecular weight.
What is identified by saponification value?
Principle: Saponification value is defined as the number of milligrams of KOH required to completely hydrolyse (saponify) one gram of the oil/fat. In practice a known amount of the oil or fat is refluxed with excess amount of standard alcoholic potash solution and the unused alkali is titrated against a standard acid.
Why do soaps need to cure?
The more important reason to cure your soap is for the water to slowly evaporate over time, which causes the soap to harden. A harder bar of soap will last longer, produce more lather, and just be an overall better bar of soap. Many soap makers let castile soap cure for six to eight months.
Is saponification a reversible reaction?
Ester hydrolysis in aqueous hydroxide is called saponification because it is used in the production of soaps from fats (Sec. 21.12B). Hence, saponification is effectively irreversible.



