What is the theory of thermal conductivity?
What is the theory of thermal conductivity?
Thermal conductivity in gases is brought about by energy transfer by gas molecules in the same way as viscosity is related with momentum transfer and diffusion, with mass transfer. Therefore, all these phenomena appreciably depend on , that is the mean free path of molecules.
What is thermal conductivity of metal rod?
Thermal Conductivity of Metal Rod: The apparatus consists of a copper bar, which is heated at one end, a heat sink is provided at other end. Heat conducted through the section of bar is measured by heat collection in water-cooled heat sink.
What are the three types of thermal conductivity?
The three types of heat transfer
- Convection.
- Conduction.
- Radiation.
What are the applications of thermal conductivity of metals?
Materials of high thermal conductivity are widely used in heat sink applications and materials of low thermal conductivity are used as thermal insulation. Thermal conductivity of materials is temperature dependent. Metals with high thermal conductivity, e.g. copper, exhibit high electrical conductivity.
How do you find thermal conductivity?
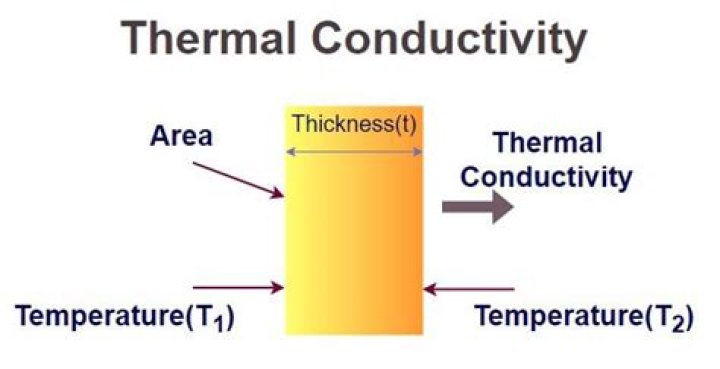
K = (QL)/(AΔT)
- K is the thermal conductivity in W/m.K.
- Q is the amount of heat transferred through the material in Joules/second or Watts.
- L is the distance between the two isothermal planes.
- A is the area of the surface in square meters.
- ΔT is the difference in temperature in Kelvin.
How do you find the thermal conductivity of a rod?
The rate of heat transfer through a rod of length x and cross-section area A whose two ends are maintained at a temperature T1 and T2 is given by dQdt=KAx(T1−T2) d Q d t = K A x ( T 1 − T 2 ) where K is the thermal conductivity of the material of the rod. The SI unit of thermal conductivity is W/(m-K).
How do you find the thermal conductivity of a metal?
What is the importance of thermal conductivity?
Thermal conductivity determines what we use to keep warm or cool and protects us from heat or cold. For example, metal is a great conductor of heat. We use it in cooking to allow heat to move through it rapidly and around what we are cooking. Other items are poor conductors of heat like rubber.
What is thermal conductivity and its examples?
Thermal conductivity is the ability of a given material to conduct or transfer heat. It is generally denoted by the symbol ‘k’ or sometimes \lambda. Materials with high thermal conductivity are used in heat sinks, on the other hand, materials with low values of \lambda used as thermal insulators.
What is metal conductivity?
Conductivity in metal is a measure of a material’s ability to transmit heat, or electricity (or sound). The reciprocal of conductivity is resistance, or the ability to reduce the flow of those. Metals conduct electricity by allowing free electrons to move between the atoms.