What isotopes go through fusion?
What isotopes go through fusion?
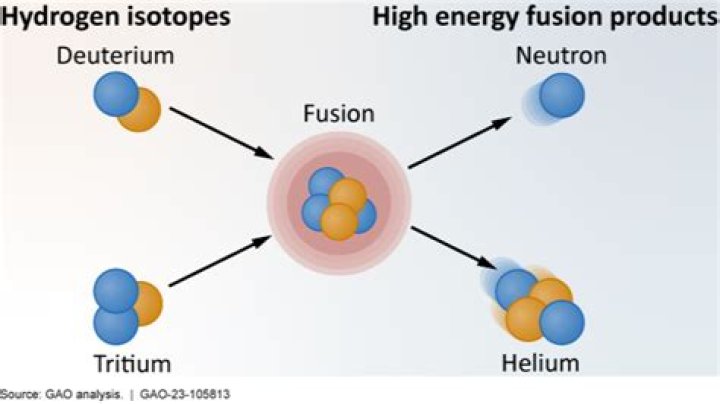
The main fuels used in nuclear fusion are deuterium and tritium, both heavy isotopes of hydrogen. Deuterium constitutes a tiny fraction of natural hydrogen, only 0,0153%, and can be extracted inexpensively from seawater.
Does fusion use hydrogen isotopes?
One possible fusion reaction occurs when different forms (or isotopes) of hydrogen collide and fuse to form nuclei of helium. These collisions also release high energy neutrons. Similar fusion reactions have been made to happen here on Earth by using the hydrogen isotopes deuterium and tritium.
Does hydrogen go through fission or fusion?
Fusion occurs when two atoms slam together to form a heavier atom, like when two hydrogen atoms fuse to form one helium atom. This is the same process that powers the sun and creates huge amounts of energy—several times greater than fission. It also doesn’t produce highly radioactive fission products.
Can hydrogen be used in fusion?
Proposed fusion reactors generally use hydrogen isotopes such as deuterium and tritium (and especially a mixture of the two), which react more easily than hydrogen to allow them to reach the Lawson criterion requirements with less extreme conditions.
Why is hydrogen used for fusion?
It takes considerable energy to force nuclei to fuse, even those of the lightest element, hydrogen. When accelerated to high enough speeds, nuclei can overcome this electrostatic repulsion and be brought close enough such that the attractive nuclear force is greater than the repulsive Coulomb force.
What are the 3 isotopes of hydrogen?
There are three isotopes of the element hydrogen: hydrogen, deuterium, and tritium. How do we distinguish between them? They each have one single proton (Z = 1), but differ in the number of their neutrons. Hydrogen has no neutron, deuterium has one, and tritium has two neutrons.
What is fusion of hydrogen?
Fusion is the process that powers the sun and the stars. It is the reaction in which two atoms of hydrogen combine together, or fuse, to form an atom of helium. In the process some of the mass of the hydrogen is converted into energy.
What is fission fusion?
Both fission and fusion are nuclear reactions that produce energy, but the processes are very different. Fission is the splitting of a heavy, unstable nucleus into two lighter nuclei, and fusion is the process where two light nuclei combine together releasing vast amounts of energy.
How many isotopes are in hydrogen?
three
Hydrogen and its two naturally occurring isotopes, deuterium and tritium. All three have the same number of protons (labeled p+) but different numbers of neutrons (labeled n).
What are 3 isotopes of oxygen?
The element oxygen has three stable isotopes: 16O, 17O, and 18O.
What are the isotopes of oxygen?
The element oxygen (O) is found in three naturally occurring stable isotopes, 18O, 17O, and 16O. The nucleus of each of these oxygen isotopes contains eight protons and either eight, nine, or ten neutrons, respectively.



