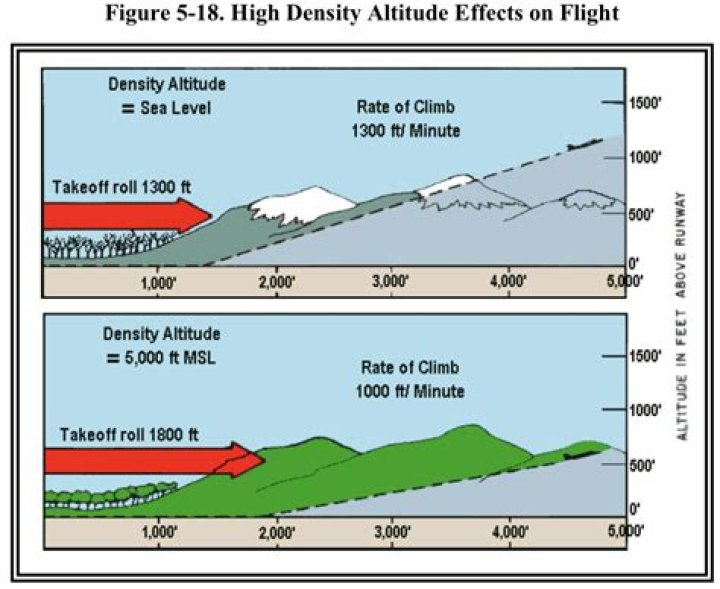
What reagent is in Sharpless Dihydroxylation?
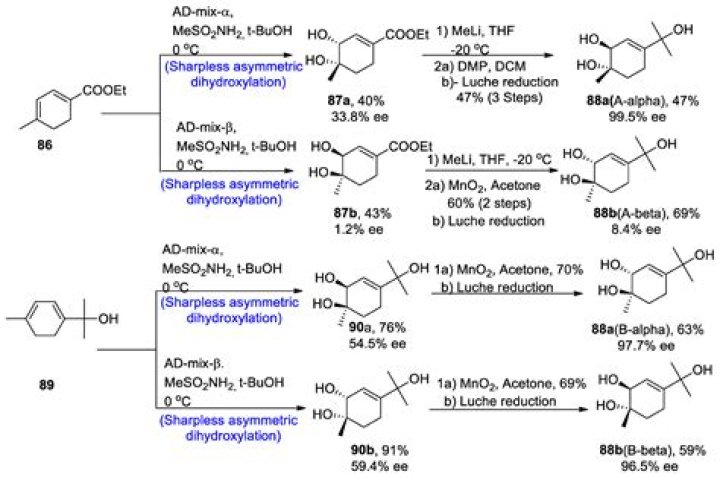
What reagent is in Sharpless Dihydroxylation?
These four reagents are commercially available premixed (“AD-mix”). The mixture containing (DHQ)2-PHAL is called AD-mix-α, and the mixture containing (DHQD)2-PHAL is called AD-mix-β….
| Sharpless asymmetric dihydroxylation | |
|---|---|
| Organic Chemistry Portal | sharpless-dihydroxylation |
| RSC ontology ID | RXNO:0000142 |
What is methanesulfonamide?
Methanesulfonamide (CH3SO2NH2) was found to be an additive that accelerates the rate-limiting hydrolysis step under the heterogeneous reaction conditions. Reaction time can be significantly shorter in the presence of CH3SO2NH2.
What is DHQD?
Hydroquinidine 1,4-phthalazinediyl diether, commonly abbreviated as (DHQD)2PHAL, is a widely used ligand for the Sharpless asymmetric dihydroxylation reaction. Nobel Prize–winning (2001) chemist K. Barry Sharpless and coauthors reviewed its use in 1994. It is sold by Sigma-Aldrich–Fluka.
What is AD mix alpha?
Reagent for Sharpless Asymmetric Dihydroxylation. AD-mix-α has been used in the asymmetric dihydroxylation (AD) step of synthesizing (+)-enantiomeric form of a potent anti-inflammatory methyl picolinate alkaloid from methyl 5-bromopicolinate.
Which is used in enantioselective preparation of 1/2 diols from Prochiral Olefines?
Sharpless Dihydroxylation
The Sharpless Dihydroxylation or Bishydroxylation is used in the enantioselective preparation of 1,2-diols from prochiral olefins.
Which reagent gives CIS diol?
o A solution of iodine and silver acetate or silver benzoate (in equimolar amount) in moist acetic acid is known as Woodward reagent o It converts the alkenes into cis-1,2 diols o In this reaction, olefin reacts with iodine in the presence of silver acetate to give an iodonium ion which undergoes displacement by …
What is Woodward reaction?
The Woodward cis-hydroxylation (also known as the Woodward reaction) is the chemical reaction of alkenes with iodine and silver acetate in wet acetic acid to form cis-diols. (conversion of olefin into cis-diol) The reaction is named after its discoverer, Robert Burns Woodward.
What is a vicinal diol?
A glycol, also known as a vicinal diol, is a compound with two -OH groups on adjacent carbons.
Is dihydroxylation an addition reaction?
Addition Reactions of Alkenes To convert alkenes into cis-diols by syn dihydroxylation, they are reacted with a basic solution of potassium permanganate (KMnO4) or Osmium tetroxide (OsO4): Both reactions go through the formation of a cyclic intermediate which is formed by a syn addition to the double bond.
What is a dihydroxylation reaction?
Dihydroxylation is the process by which an alkene is converted into a vicinal diol. Although there are many routes to accomplish this oxidation, the most common and direct processes use a high-oxidation-state transition metal (typically osmium or manganese).
What is SYN Dihydroxylation?
Reactions that add two hydroxyls to the same face of an alkene double bond as it’s converted to a single bond.