What type of enzyme is trypsin?
What type of enzyme is trypsin?
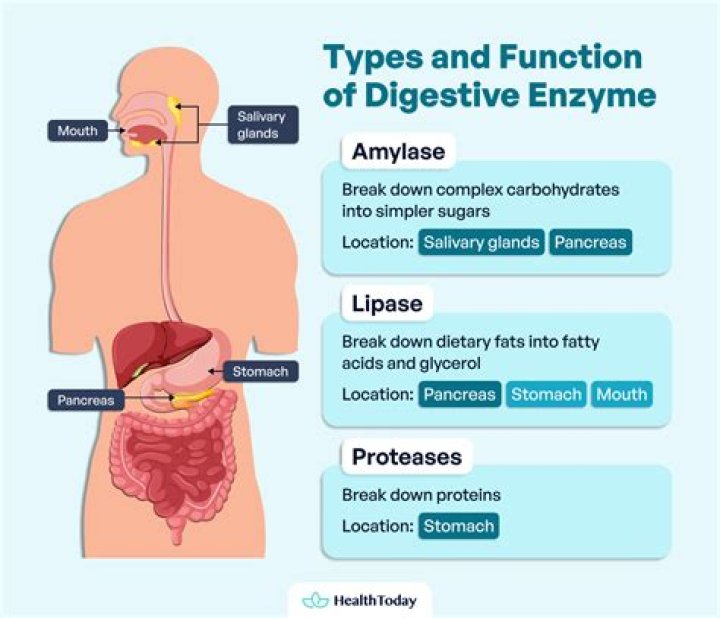
proteolytic enzyme
Trypsin is an enzyme that helps us digest protein. In the small intestine, trypsin breaks down proteins, continuing the process of digestion that began in the stomach. It may also be referred to as a proteolytic enzyme, or proteinase. Trypsin is produced by the pancreas in an inactive form called trypsinogen.
What type of protease is trypsin?
serine protease
Trypsin is a serine protease found in the digestive system of many vertebrates, where it hydrolyzes proteins at the carboxyl side of the amino acids lysine or arginine.
Why is trypsin used in mass spectrometry?
Trypsin is the most popular protease used in mass spectrometry because of its high proteolytic activity and cleavage specificity. Trypsin and Lys-C work simultaneously to digest proteins under conventional non-denaturing conditions and improve trypsin digestion without procedural changes.
What is the active form of Chymotrypsinogen?
trypsin
Chymotrypsinogen must be inactive until it gets to the digestive tract. This prevents damage to the pancreas or any other organs. It is activated into its active form by another enzyme called trypsin. This active form is called π-chymotrypsin and is used to create α-chymotrypsin.
What is trypsin Class 10?
Trypsin, a serine protease is an enzyme that helps us in digesting protein. It continues the process of digestion that began in the stomach in the small intestine by breaking down proteins. This enzyme is produced by the pancreas in an inactive form called trypsinogen.
What enzymes are activated by trypsin?
Trypsinogen is activated by enterokinase, which cleaves an amino-terminal activation peptide (TAP). Active trypsin then cleaves and activates all of the other pancreatic proteases, a phospholipase, and colipase, which is necessary for the physiological action of pancreatic triglyceride lipase.
How trypsin acts on a peptide?
Function. In the duodenum, trypsin catalyzes the hydrolysis of peptide bonds, breaking down proteins into smaller peptides. The peptide products are then further hydrolyzed into amino acids via other proteases, rendering them available for absorption into the blood stream.
Why is trypsin a serine protease?
Trypsin is a medium size globular protein that functions as a pancreatic serine protease. This enzyme hydrolyzes bonds by cleaving peptides on the C-terminal side of the amino acid residues lysine and arginine. It cleaves linkages involving lysine and arginine.
Is trypsin a serine protease?
Clan PA proteases bearing the trypsin fold are the largest family of serine proteases and perhaps the best studied group of enzymes. Digestive enzymes such as trypsin and chymotrypsin cleave polypeptide chains at positively charged (Arg/Lys) or large hydrophobic (Phe/Trp/Tyr) residues, respectively.
Why trypsin is used in proteomics?
Trypsin is the protease of choice for mass spectrometry (MS)-based proteomics. It cleaves carboxyterminal of Arg and Lys residues, resulting in a positive charge at the peptide C-terminus, which is advantageous for MS analysis. In contrast, broad specificity proteases are much less widely used in proteomics.
How does trypsin work?
In the duodenum, trypsin catalyzes the hydrolysis of peptide bonds, breaking down proteins into smaller peptides. The peptide products are then further hydrolyzed into amino acids via other proteases, rendering them available for absorption into the blood stream.



