Which family of elements has 4 valence electrons?
Which family of elements has 4 valence electrons?
Carbon Group
Carbon Group or Tetrels: – Group 14 (IVA) – 4 valence electrons.
Does the carbon family have 4 valence electrons?
All the carbon group atoms, having four valence electrons, form covalent bonds with nonmetal atoms; carbon and silicon cannot lose or gain electrons to form free ions, whereas germanium, tin, and lead do form metallic ions but only with two positive charges.
What element is in the carbon family Period 4?
Group 4A (or IVA) of the periodic table includes the nonmetal carbon (C), the metalloids silicon (Si) and germanium (Ge), the metals tin (Sn) and lead (Pb), and the yet-unnamed artificially-produced element ununquadium (Uuq).
What is the number of valence electrons for carbon Group 4?
4 valence electrons
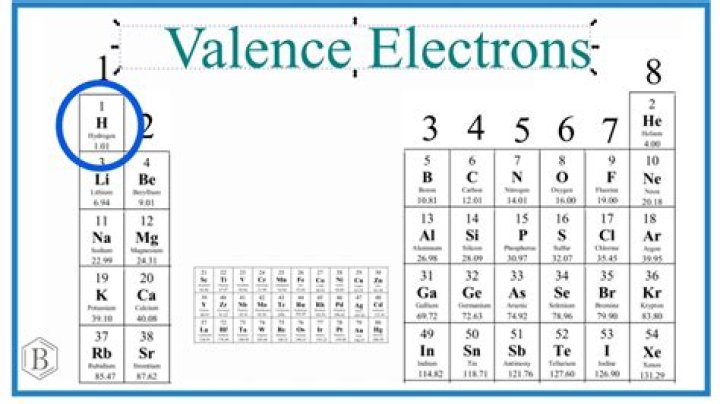
Look at the writing above each Group, or column. The number next to the “A” is the number of valence electrons in an atom of an element in that Group. Carbon is in Group 4A, so it has 4 valence electrons.
Does be have 4 valence electrons?
An atom with a closed shell of valence electrons (corresponding to an electron configuration s2p6) tends to be chemically inert….The number of valence electrons.
| Periodic table group | Valence Electrons |
|---|---|
| Group 14 (IV) (carbon group) | 4 |
| Group 15 (V) (pnictogens) | 5 |
| Group 16 (VI) (chalcogens) | 6 |
| Group 17 (VII) (halogens) | 7 |
Which of the following has exactly 4 valence electrons?
Carbon, silicon and germanium are semiconductor elements, and these have precisely four valence electrons in their atoms.
What element is in Group 4 period 4?
titanium
Group 4 is the second group of transition metals in the periodic table. It contains the four elements titanium (Ti), zirconium (Zr), hafnium (Hf), and rutherfordium (Rf). The group is also called the titanium group or titanium family after its lightest member….Group 4 element.
| Titanium |
| Zinc |
| Gallium |
| Germanium |
| Arsenic |
Do Group 4 elements form ions?
None of the Group 4 elements form 1+ ions, so looking at the first ionization energy alone is not helpful. Some of the elements do, however, form 2+ and (to some extent) 4+ ions. The first chart shows how the total ionization energy needed to form the 2+ ions varies down the group.
What element is in Group 4 Period 4?
What element has 4 protons 4 electrons and 6 neutrons?
Beryllium
Atomic Number
| Name | Protons | Neutrons |
|---|---|---|
| Lithium | 3 | 4 |
| Beryllium | 4 | 5 |
| Boron | 5 | 6 |
| Carbon | 6 | 6 |
Does potassium have 4 valence electrons?
K is the symbol for potassium, and the number of valence electron can be found through its’ group on the periodic table. Hence, it has one valence electron.
How many of valence electrons do the elements in column 4 have are these elements very reactive explain your answer?
Explanation: Elements in group 14 have 4 valence electrons. Meaning that to achieve a stable octet they need to either gain or lose 4 electrons in total.