Which is favored keto or enol?
Which is favored keto or enol?
Phenols can theoretically exist in their keto forms, but the enol form is greatly favored due to aromatic stabilization.
What is the keto form of glucose?
Glucose and galactose are aldoses. Fructose is a ketose. Monosaccharides can exist as a linear chain or as ring-shaped molecules; in aqueous solutions they are usually found in ring forms (Figure 3).
What are 2 examples of tautomers for D fructose?
The minor tautomers of fructose are α-D-fructopyranose (α-pyr) and the linear keto form of fructose.
Why is keto more stable than enol form?
In most keto-enol tautomerisms, the equilibrium lies by far toward the keto form, indicating that the keto form is usually much more stable than the enol form, which can be attributed to the feet that a carbon-oxygen double bond is significantly stronger than a carbon-carbon double bond.
Why is enol less stable than keto?
In general, enols are less stable than their keto equivalents, because of the favorability of the C=O. double bond over C=C double bond. However, enols can be stabilized kinetically or thermodynamically.
When is enol more stable than keto?
In general , keto form is more stable than enol form due to greater bond energy of c=o. than c=c . But, in some cases enol form is more stable than keto . For example, if bulky phenyl groups are present at sp3 alpha carbon then enol is more stable than keto form due greater bond angle.
What is the difference between enol and enolate?
An enolate is the conjugated base of an enol. The key difference between enols, enolates and enamines is that enols contain a hydroxyl group with an adjacent C=C double bond and enolates contain a negative charge on the oxygen atom of an enol whereas enamines contain an amine group adjacent to a C=C double bond.
What is nutritional ketosis level?
Nutritional ketosis is defined as blood ketone levels of 0.5 – 3 mmol/L, which also represents the optimal ketone range for weight loss. To achieve these levels, you need to significantly restrict your carb intake.
What do you mean by Keto-enol tautomerism?
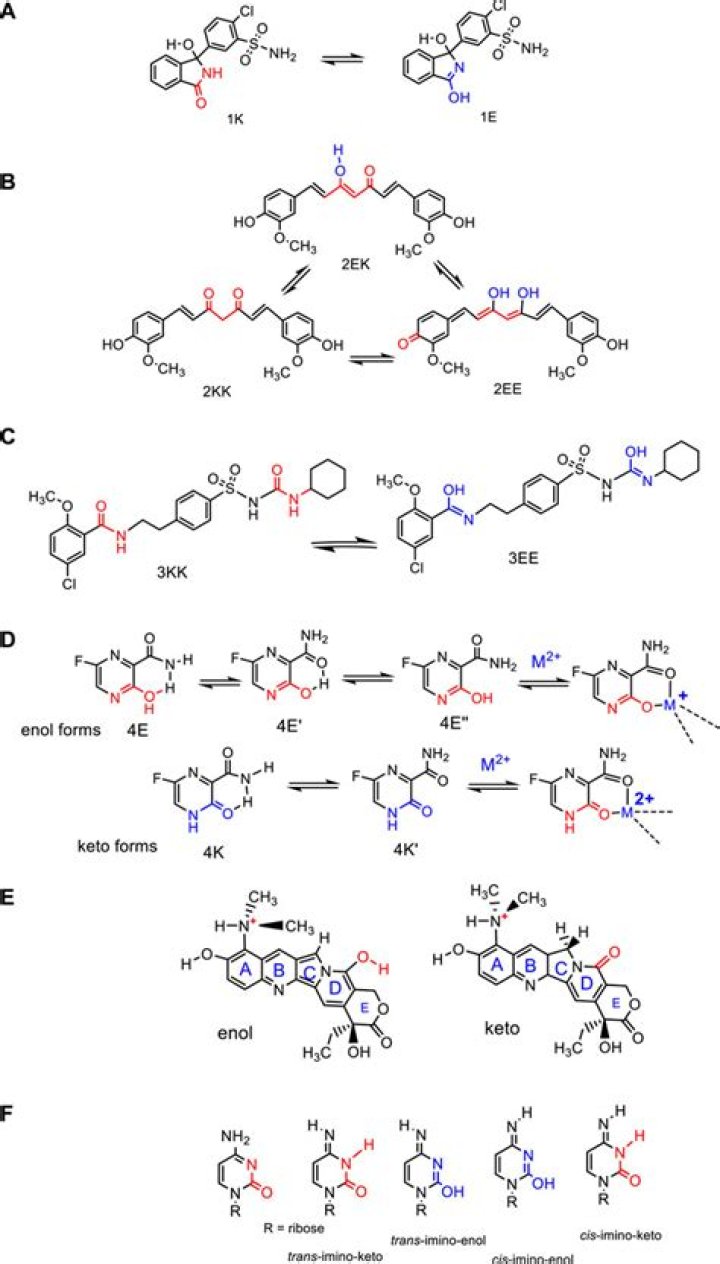
Keto-enol tautomerism refers to a chemical equilibrium between the keto form (carbonyl structure containing α-hydrogen) and the enol form (a double bond adjacent to an alcohol, -C=C-OH) of a compound. Its stability depends upon factors like aromaticity, hydrogen bonding, solvent, conjugation, and substitution [1-5].
In which one of the following keto-enol systems the keto form is more stable than the enol form?
Enolic form of phenol is more stable than keto form by -13 kcal/mole of energy hence phenol exist exclusively as an enol. Enolic tautomer is less polar due to intramolecular hydrogen bonds than the corresponding keto form.