Why are liquids able to flow while solids are not
Because the particles can move, liquids don’t have a definite shape, and they can flow. Because the particles are still packed close together, liquids can’t easily be compressed and keep the same volume.
Why do liquids flow but solids do not?
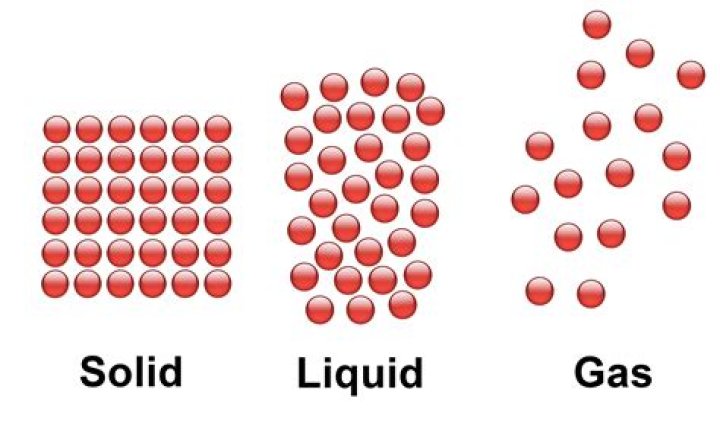
(a) The molecules of liquids and gases are far apart i.e. have more gaps, intermolecular attraction force is very less as compared to solids, hence liquids and gases can flow but solids do not as gaps*in solid molecules is less and molecular force of attraction very strong.
Why do solid objects not flow?
Unlike a liquid, a solid object does not flow to take on the shape of its container, nor does it expand to fill the entire available volume like a gas. … Solids cannot be compressed with little pressure whereas gases can be compressed with little pressure because the molecules in a gas are loosely packed.
Why can liquids easily flow?
A liquid generally flows easily because the intermolecular space between the liquid particles are more as compared to solids. Moreover there particles are loosely packed due to the presence of a little force of attraction between them.Why do solids flow?
Solid matter (such as sand) that is made up of many individual small particles is called a granular material, and the individual particles are called grains. … In these factories granular materials usually flow out of a large container called a hopper and through a funnel.
Why can liquids flow whereas solids can be heaped?
Liquids always tend to flow, because the intermolecular forces between molecules is less. However in solids, the bonds are very strong and prevent molecules from moving around. This makes it easier for solids to be heaped, without losing their shape and structure.
Why can't solids and liquids be compressed?
If we put pressure on a solid or a liquid, there is essentially no change in volume. The atoms, ions, or molecules that make up the solid or liquid are very close together. There is no space between the individual particles, so they cannot pack together.
Do liquids flow?
Liquids will flow and fill the lowest portion of a container, taking on the shape of the container but not changing in volume. The limited amount of space between particles means that liquids have only very limited compressibility.Why are liquids more fluid than solids?
Liquids and gases are considered to be fluids because they yield to shearing forces, whereas solids resist them. … This rock retains its shape because of the forces holding its atoms together. (b) Atoms in a liquid are also in close contact but can slide over one another.
Can a solids flow?Granular materials are solids that can flow and behave like liquids. Examples of granular materials include sand, coffee, nuts, even corn flakes. Scientists consider things like icebergs or asteroids to be large granular materials, due to how they behave.
Article first time published onWhy liquids can be compressed?
In most liquids, the particles are less densely packed, so they can move around and slide past each other. While a liquid is easier to compress than a solid, it is still quite difficult – imagine trying to compress water in a container.
What is the flow of solids?
There are several modes of solids flow in a hopper: funnel flow, mass flow, etc. Funnel flow would have an annular zone of stationary solids and a moving core of solids at the center. In this case, the solids flow primarily through the core of the hopper. Solids in the periphery either remain stationary (Fig.
Why do liquids and gases flow?
Liquids, solids, and gases. Liquids flow because the intermolecular forces between molecules are weak enough to allow the molecules to move around relative to one another. Intermolecular forces are the forces between neighboring molecules. … First, liquids share the ability to flow with gases.
What flow do solids have?
Solids stay in one place and can be held. Solids keep their shape. They do not flow like liquids. Solids always take up the same amount of space.
Why are solids and liquids harder to compress than gases?
In a liquid the particles are still close together but a little further apart than in a solid. … There is still very little space between particles, so liquids are also difficult to compress. Gases. In gases the particles are much further apart than in solids or liquids.
Why do solids and liquids behave differently?
Liquids differ from solids in that the heat levels (molecular vibration) in the molecules of the substance are sufficient to allow them to flow past one another without forming a rigid bond structure. In this state of matter, the molecules exert force downward and outward (toward the sides of its container).
Why are solids and liquids incompressible while gases are compressible?
Why are solids and liquids incompressible while gases are compressible? Solids form closed packed structure with negligible intermolecular space. … Liquids have very less intermolecular spaces so even they are resistant to external pressure and does not change their shape hence are incompressible.
Why do solids not flow Class 4?
SolidsEdit They are held tightly together by strong forces of attraction. They are held in fixed positions but they do vibrate. Because the particles don’t move, solids have a definite shape and volume, and can’t flow.
Why a liquid can flow and it needs a vessel to contain?
Answer: The fluidity of the liquid state is because the constituent particles are loosely bound with one another or the intermolecular forces are weak due to which the particles can move freely but not as freely as they do in gas phase so in order to contain them you need to keep them bound.
Why solid has definite volume and definite shape?
(a) Solids have definite shape and definite volume because the molecules in solid are closely packed and in fixed positions. The molecules can vibrate but do not move around which keeps the shape and volume definite.
Why do liquid and gas particles can move freely?
The attractive forces between particles are strong enough to hold a specific volume but not strong enough to keep the molecules sliding over each other. … The kinetic energy of the molecule is greater than the attractive force between them, thus they are much farther apart and move freely of each other.
Why do solids liquids and gases differ in their physical state?
Solids, liquids and gases differ in their physical state due to the difference in the intermolecular force of attraction between the particles of each of them. The force of attraction in the case of solids is highest whereas it is lower in liquids and further lowest in gases.
Why are liquids called fluids?
Liquids and gases are called fluids because they can be made to flow, or move. In any fluid, the molecules themselves are in constant, random motion, colliding with each other and with the walls of any container. … Under standard atmospheric conditions, water exists as a liquid.
Do all liquids flow freely?
When you pour a glass of water, or fill a car with gasoline, you observe that water and gasoline flow freely. … Water, gasoline, and other liquids that flow freely have a low viscosity. Honey, syrup, motor oil, and other liquids that do not flow freely, like those shown in Figure 1, have higher viscosities.
Can solids diffuse into solids?
Answer: Yes solid can diffuse with solid an example for this is copper+ gold together can be diffuse together….
Why sand can flow like a liquid yet it is not a liquid?
Ans: Sand can flow like a liquid,yet it is not a liquid because both sand and cereal are granular materials. That means they are made up of solid particles,but they can actually flow like liquids.
What can not flow?
Solids: have a fixed shape and cannot flow, because their particles cannot move from place to place. cannot be compressed (squashed), because their particles are close together and have no space to move into.
What are the various factors affecting the solid flow?
When handling bulk solids, there are several factors that can affect your material flow. Most of these are environmental; all of them need to be taken into consideration. Moisture content, temperature, particle size, and time of storage at rest can have a tremendous impact on your material’s flow properties.
Can liquids be compressed into solids?
Liquids are compressible. But their compression rate is very less. The atoms in it are packed little loosely. For solids its tight so solids cannot be compressed.
Can solids be compressed?
The particles in solids are very close together, therefore they cannot usually be compressed or squashed. Forces of attraction between the particles hold them together and keep them in place. The particles in solids are arranged in a regular way. The particles in solids move only by vibrating about a fixed position.
Why can gases flow easily?
Particles of gas move freely as there is lot of space and they have more kinetic energy than liquids and solids. Gases have the highest rate of diffusion.