Why are radioactive isotopes useful in scientific research?
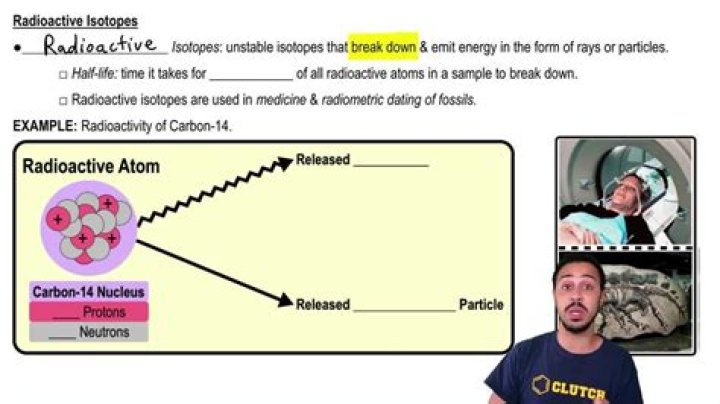
Why are radioactive isotopes useful in scientific research?
Radioactive isotopes are effective tracers because their radioactivity is easy to detect. A tracer is a substance that can be used to follow the pathway of that substance through some structure.
How is half-life useful to scientists?
Scientists look at half-life decay rates of radioactive isotopes to estimate when a particular atom might decay. A useful application of half-lives is radioactive dating. This has to do with figuring out the age of ancient things. It takes a certain amount of time for half the atoms in a sample to decay.
How can isotopes and half-lives be useful?
They allow us to determine the ages of very old artifacts. Scientists can use the half-life of Carbon-14 to determine the approximate age of organic objects less than 40,000 years old. Isotopes with longer half-lives such as Uranium-238 can be used to date even older objects.
How are radioactive isotopes used in science and industry?
Radioisotopes are used by manufacturers as tracers to monitor fluid flow and filtration, detect leaks, and gauge engine wear and corrosion of process equipment. Radiotracers are also used in the oil and gas industry to help determine the extent of oil fields.
What are the four uses of radioactive isotopes?
Different chemical forms are used for brain, bone, liver, spleen and kidney imaging and also for blood flow studies. Used to locate leaks in industrial pipe lines…and in oil well studies. Used in nuclear medicine for nuclear cardiology and tumor detection. Used to study bone formation and metabolism.
How is the half-life of a radioactive material useful for radioactive dating?
The half-life of an isotope is used to describe the rate at which the isotope will decay and give off radiation. Using the half-life, it is possible to predict the amount of radioactive material that will remain after a given amount of time.
Why do scientists use half-life to describe radioactive decay explain?
By comparing the normal abundance of carbon-14 in a living creature (which is the same concentration in the atmosphere) with the amount left in the material being dated, based on the known decay rate, scientists can figure out roughly how long ago whatever they’re looking at was still alive.
What is half-life of a radioactive isotope?
half-life, in radioactivity, the interval of time required for one-half of the atomic nuclei of a radioactive sample to decay (change spontaneously into other nuclear species by emitting particles and energy), or, equivalently, the time interval required for the number of disintegrations per second of a radioactive …
What is meant by the half-life of a radioactive isotope?
What is half-life radioactivity write some applications of radioisotopes in biological sciences?
This decaying property of radioisotopes is called half-life. Thus radioisotopes could be used for numerous biomedical purposes such as cancer and tumour treatment, imaging, biochemical assays, biological labelling, sterilization, clinical diagnostics, radioactive dating etc.
What are 3 uses of radioactive isotopes?
Used in cancer treatment, food irradiation, gauges, and radiography.
What is a half-life of radioactive material?
Half-life is the length of time it takes for half of the radioactive atoms of a specific radionuclide to decay. A good rule of thumb is that, after seven half-lives, you will have less than one percent of the original amount of radiation.